The way nuclear weapons work is in one sense fairly simple, but the details are pretty complicated. This page provides some information to help you understand the trickier parts of nuclear weapons functioning. There is nothing secret here - at least not today. Fifty years ago some of this was classified Top Secret Restricted Nuclear Weapons Design Information. It is all basic physics. I have simplified it quite a bit, leaving out details that are not essential to understanding how the bomb works.
Radioactivity
Nuclear weapons work by breaking apart atoms. Matter is made up of atoms. These come in many varieties called elements. Aluminum, iron, carbon, oxygen and hydrogen are a few of the elements. They are different because their atoms are composed of different numbers of the basic atomic building blocks - protons, neutrons and electrons. At the center of an atom is a nucleus, where the protons and neutrons are, and around the nucleus are the electrons.
The type of element is determined by the number of protons in the nucleus. Hydrogen has 1 proton, oxygen has 8, iron has 26 and lead has 82. The number of neutrons is about the same as protons, but can vary quite a bit. For example, oxygen can have from 6 to 12 neutrons. Each of these different combinations of protons and neutrons is called an isotope.
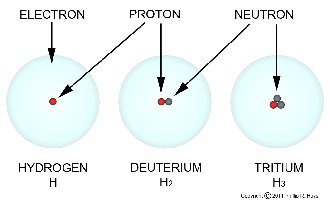
The different isotopes of elements are shown as the symbol of the element followed by the sum of protons and neutrons (the atomic weight of the element). For example the three isotopes of hydrogen are H1, H2 and H3. H1 is ordinary hydrogen and is usually just written as H. H2 is deuterium and H3 is tritium (note that most isotopes of elements do not have common names, just numbers). The isotopes of uranium are U227 through U240, and U234, U235 and U238 are the naturally occurring isotopes on Earth (99.27% is U238).
While it is possible for an element to have many isotopes, some or all of them may be unstable. Stable isotopes have nuclei that do not change. They make up most of the things that surround us. Once created these atoms continue to exist for very long times, perhaps as long as the universe itself. Unstable isotopes have weaker forces binding the protons and neutrons together in their nuclei. Occasionally something happens to rearrange the components of the nucleus. The result is the creation of a new nucleus and emission of radiation, usually in the form of alpha particles, beta particles or gamma rays. These unstable isotopes are called radioisotopes. This radioactive decay usually produces a new element with one or two fewer protons than the original radioisotope.
The isotopes of oxygen are O14 through O20. All have 8 protons but the number of neutrons varies from 6 to 12. Oxygen has three stable isotopes, with 8, 9 and 10 neutrons. Although all three are naturally occurring and stable, on Earth 99.759% of oxygen atoms have eight neutrons (O16). The four unstable oxygen radioisotopes do not exist naturally, and are produced on Earth only in nuclear reactors and nuclear explosions. They decay rapidly and emit both beta and gamma radiation.
Radioactive decay of individual atoms is unpredictable. Even though an isotope is unstable, a particular nucleus may decay immediately after it is formed, or may hang around for quite a while. However, the behavior of a large number of nuclei of the same isotope is quite predictable. Within a large population some nuclei decay quickly, others more slowly, but the average decay time will be some where in between. We cannot say how long it will take for all atoms to decay but we can predict how long it will take for some of them to decay.
We can perform experiments to determine how long it will take for half of the atoms to decay. This time period is called the half life of the isotope. For example, the half lives of the four unstable oxygen isotopes are 71, 124, 29 and 14 seconds. In this last case, in 14 seconds half of the nuclei will have decayed. In another 14 seconds half of the remaining nuclei will decay, and so on until eventually none remain. With the longest oxygen radioisotope half life being only about two minutes, virtually none of these radioisotopes will be around a day after they are created.
There are 92 naturally occurring elements on Earth, and all of them have multiple isotopes. Hydrogen is the lightest element, with 1 proton and three isotopes with 0, 1 and 2 neutrons. Uranium is the heaviest naturally occurring element with 93 protons and from 135 to 148 neutrons. None of the uranium isotopes are stable, and only three are naturally occurring. The naturally occurring isotopes have half lives of from 25 million years to 4.5 billion years. So, although these isotopes are unstable, because they have very long half lives there still is quite a bit of uranium on Earth. The uranium isotopes that do not occur naturally have much shorter half lives, most measured in days, hours or minutes.
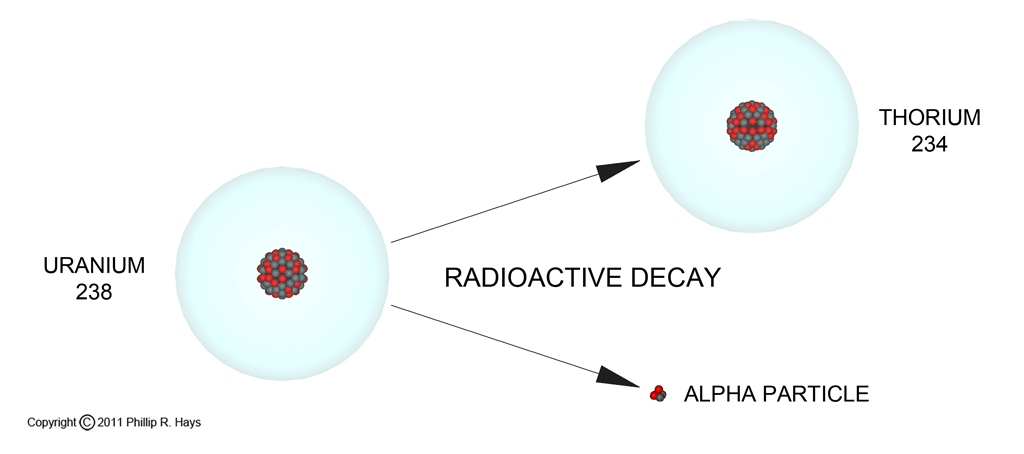
Normal radioactive decay of uranium occurs by emission of an alpha particle (alpha radiation) from the nucleus. An alpha particle is composed of two protons and two neutrons. When the nucleus loses an alpha particle it loses two protons and becomes another element. For example, when U238 decays, in addition to the alpha particle a new nucleus of the element thorium (Th234) is produced. The thorium atom is the decay product of the reaction. The sum of the protons and neutrons in the alpha particle and Thorium atom equals the number of particles in the original uranium atom (4 + 234 = 238).
Nuclear Fission
In addition to simple radioactive decay some elements undergo nuclear fission where the nucleus disintegrates into a collection of particles and smaller nuclei (fission products). When this happens a lot of energy is released, and this is where nuclear weapons get their BANG! Uranium atoms occasionally disintegrate to produce two much smaller nuclei, a shower of particles and gamma radiation. However, spontaneous fission occurs relatively infrequently. In a gram of U235 there are 2,560,000,000,000,000,000,000 atoms, but only eight spontaneous fissions occur in an hour, or one every 7.4 minutes.
In the mid 1930s some German physicists discovered that it is possible to cause uranium nuclei to break up pretty much on command. So instead of having to wait for uranium nuclei to split, it is possible make them break up when you want them to. All you have to do is hit them with extra neutrons and the nuclei become more unstable, with very short half lives.
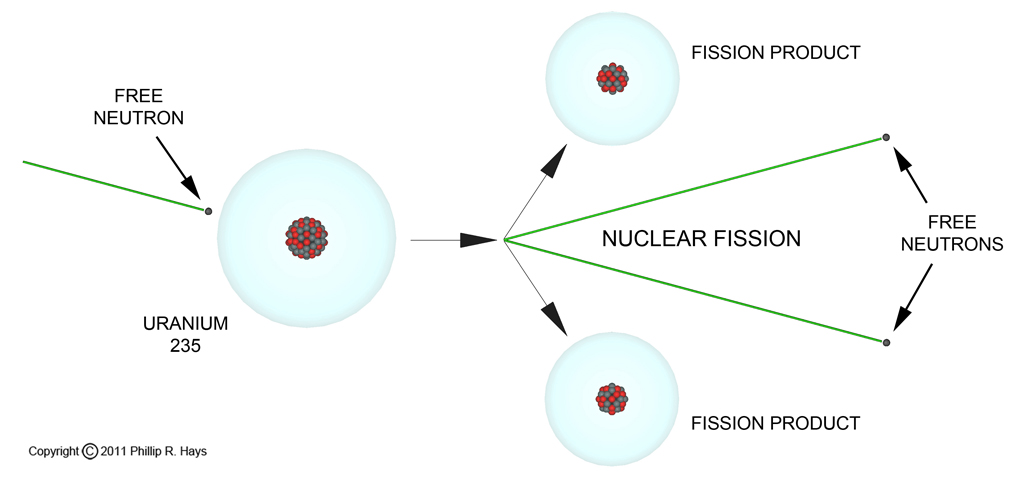
Even more interesting is the fact that when a U235 nucleus fissions it spits out from one to five neutrons (there are about 40 ways uranium atoms can fission). Each of these free neutrons can then cause another uranium nucleus to fission, and each of these releases more neutrons in a chain reaction. So one neutron becomes two, and these become four, then eight, sixteen, and so on (each stage of the chain reaction is called a generation). The average time between fission generations is about 10 nanoseconds (0.00000001 second). Very quickly an exponentially increasing flood of neutrons is released, causing billions of nuclei to fission in a very short time, releasing huge amounts of energy. Nuclear energy, in a nuclear explosion.
Actually, it isn't quite that simple. Virtually all of the uranium on Earth is U238, and it is not easy to cause it to undergo this nuclear chain reaction. The isotopes U234 and U235 do fission quite easily if struck by neutrons of proper energy, and they conveniently liberate more neutrons with just the right energy to sustain a chain reaction. However, these isotopes are very scarce and it is difficult to get enough of these materials to make a bomb - but that is another story.
Suppose you can produce U235, the second most abundant uranium isotope on Earth, how much do you need to make a bomb? That is actually pretty simple. But first a few words about atoms. Atoms are mostly empty space, with the relatively tiny nucleus surrounded by a large electron cloud. Consequently, any free neutron passing by is most likely to pass right through the atom without striking the nucleus. No collision, no fission.
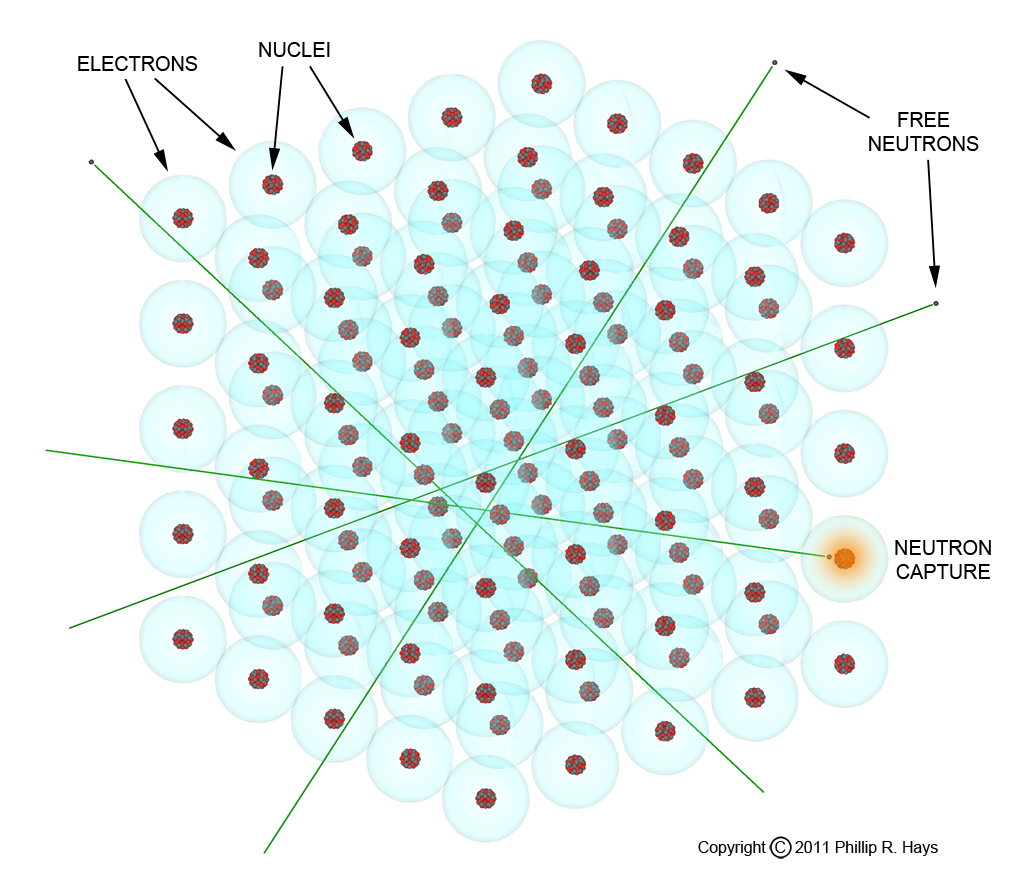
But if you have enough atoms crammed closely together, eventually a free neutron will strike a nucleus (neutron capture) and cause it to fission. Of course this is problematic, because the neutron may also pass all the way through the material without striking anything and escape elsewhere. The larger the piece of fissile material you have the greater the probability that a passing neutron will collide with a nucleus. Physicists have created the term mean free path to describe just how much fissile material a neutron must pass through to get a high probability that it will be captured and cause a nucleus to fission. The optimal shape for the fissionable material is spherical, with the greatest amount of material in the smallest possible volume.
For a nuclear chain reaction to occur at least one of the neutrons generated by each splitting nucleus must be captured and cause another nucleus to fission. For this to occur the diameter of the fissionable material must be greater than or equal to the mean free path. If the diameter is too small a chain reaction will not be sustained (subcritical) and the rate of neutron production will be just the natural spontaneous rate of decay of the isotope. If the diameter is equal to the mean free path the rate of neutron capture will be sufficient to maintain a non-increasing chain reaction - this is a critical mass, and it is the basis of nuclear reactors that produce a continuous amount of energy. If the diameter is greater than the mean free path the chain reaction will grow exponentially (supercritical) and a nuclear explosion will occur.
For U235 a critical mass is 52 Kg and it has a diameter of 16.5 cm. For the plutonium isotope Pu239 the critical mass is 10 Kg and the diameter is 9.9 cm. You can see why plutonium is a more favorable bomb material - the weapon can be made smaller and lighter weight.
Assembly
So all you need to do to produce a nuclear explosion is to assemble a supercritical mass of a spontaneously fissioning isotope. Right? It sounds simple, but the trick is to assemble it at the correct time. You can't just dump a supercritical mass of U235 into a can and haul it around until you want an explosion, because it might go off at any time, and a nuclear explosion at the wrong time will ruin your entire day.
The simplest way to assemble a uranium bomb is to separate the supercritical mass into two subcritical parts. These will sit around forever without exploding. Then, when you want an explosion you just bring the two parts together and wait for a spontaneous fission event to get things started. However, you must bring the parts together very quickly because there may be free neutrons leaking out of each subcritical piece and as the two pieces come together these neutrons will start interacting with nuclei to increase the fission rate. This liberates heat, and the whole thing could melt or be blown apart in a small explosion caused by the sudden release of heat before a supercritical reaction can start. The common way to avoid this problem is to bring the two subcritical masses together extremely quickly before they can heat up. One piece is fixed in place and the other is accelerated down a gun barrel by an ordinary high explosive charge. The two pieces slam together at high speed and form a supercritical mass. This was the mechanism for the "Little Boy" bomb that was dropped on Hiroshima. One nice thing about this method is that it is very simple and cannot fail to work.
However, the gun mechanism is not very efficient. It requires a relatively large amount of fissile material to ensure that the chain reaction will start (greater weight and higher cost). In addition, only a small amount of the fissionable material actually fissions, causing a relatively low yield. Only 1.4% of the uranium fissioned in the Little Boy bomb. Also, a gun-type mechanism will not work with the Pu239 isotope of plutonium that is used in nuclear weapons. All plutonium contains a small amount of the Pu240 isotope that is very unstable. A stray neutron will produce a supercritical chain reaction much more quickly in plutonium. Two masses of plutonium cannot be brought together fast enough in a gun type mechanism to prevent premature detonation.
Another way to assemble a supercritical mass is to form the fissile material into a hollow sphere where the thickness of the shell is less than the mean free path. In this way fissile material can be formed into a subcritical shell larger in diameter than the critical size, and then can be squashed into a smaller supercritical mass in a very short time. This hollow shell of fissile material is called a pit - like the pit of a cherry. All you need is a spherical implosion. This is easier to describe than to achieve, but it is the way many nuclear weapons work, including the "Fat Man" bomb that was dropped on Nagasaki, and the W-30 warhead of the Talos missile.
An implosion system that is just made of a layer of high explosives that surrounds the hollow fissile material will not work. The problem is that explosions normally radiate outward through the explosives in all directions from the point of detonation. Such a wave will strike one point on the surface of the pit first, pushing the material at the first point of contact inward and deforming the pit. The pit will not form a uniform highly compressed sphere with the proper dimensions. If this happens the fissile material will not go supercritical and generate a full scale nuclear explosion.
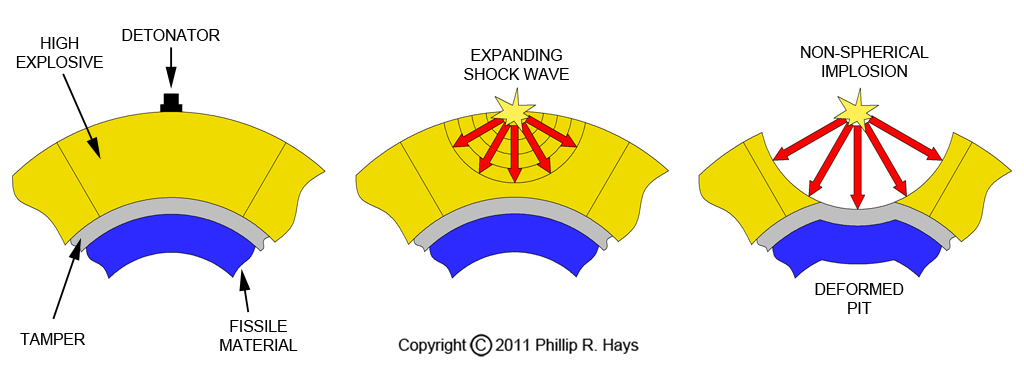
What is needed for an implosion bomb is a way to reshape the shock wave so it strikes all parts of the surface of the pit at the same time. This is accomplished by using an explosive lens system. The high explosives used in nuclear weapons do not actually burn like gunpowder and other ordinary explosives. Instead a shock wave passes through the material detonating the explosive as it passes. The rate the detonation wave moves through the material ranges from about 5000 to 10,000 meters per second, depending upon the chemical composition of the explosives. Nuclear weapons use specially designed plastic explosives with carefully controlled detonation rates.
On the outside of the lens assembly is the detonator. It is attached to a mass of explosive that detonates at high velocity (yellow). Inside the high velocity explosive is another explosive that detonates at a lower velocity (orange). These two masses of explosive are shaped to refract the shock wave similar to the way that optical lenses are designed to refract light. The inner surface of the low velocity explosive is spherical and rests against the spherical pit. The surface between the two explosive types is more or less conical, but instead of coming to a sharp point it is rounded. The shape of this surface is calculated based upon the relative detonation rates of the two explosives. The explosive lens system focuses the energy of the chemical explosion on the surface of the pit.
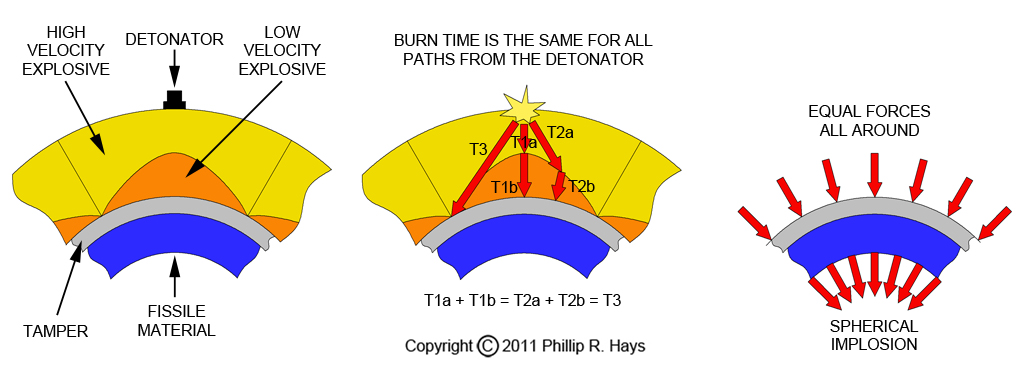
The explosion shock wave passes quickly through the high velocity explosive until it contacts the low velocity explosive. There the shock detonates the low velocity explosive. The detonation wave moves more slowly through the low velocity explosive. The shock wave arrives at points on the boundary between the two explosives at different times, arriving first at the point closest to the detonator and last at the point most distant. Along the shortest direct path from the detonator to the surface of the pit there is more low velocity explosive, delaying the shock wave as it expands inward. Along the longer paths from the detonator to the pit there is more high velocity explosive to allow the wave to traverse the longer distance faster. The amounts of fast and slow explosives are calculated to shape the shock wave so it travels along all paths from the detonator to the surface of the pit in the same amount of time (about 10 to 15 microseconds). This causes the shock wave to be shaped into a spherical implosion that arrives at all points on the surface of the pit at the same instant. With all forces equal all around, the pit is compressed uniformly into a compact high density sphere to form a supercritical mass. The creation of this explosive lens system is one of the major tasks in the design of implosion bombs.
The implosion wave crushes the hollow pit into a small supercritical ball. The pressure on the pit material can be several million times atmospheric pressure. This exerts a force so great that the atoms in the fissile material are compressed to two to three times normal density. This causes the distance between atoms to be reduced significantly, reducing the mean free path and increasing the probability of neutron capture. Reduction of the mean free path also reduces the time between fission generations, speeding up the chain reaction. A relatively small subcritical amount of fissile material can be compressed to make it supercritical, allowing a smaller, lighter weight and less expensive amount of uranium or plutonium to be used in an implosion weapon.
The implosion takes much less than a millisecond to achieve supercriticality. To illustrate the extremely brief duration of the nuclear explosion, from the moment the detonators fire the shock wave from the high explosives travels outward from the surface of the explosives through air (at the speed of sound) only about half an inch (1 centimeter) before the supercritical nuclear reaction starts. Before the blast from the chemical explosives can break out of the warhead shell the entire warhead assembly is vaporized by the initial burst of radiation from the nuclear explosion.
Just creating the supercritical mass is not enough to ensure that a nuclear explosion will occur, or that it will produce the desired yield. A basic implosion assembly can result in about 20% of the fissile material undergoing fission. Several other features are designed into nuclear weapons to enhance yield and reliability.
Neutron Generators
Now suppose you have figured out how to make an implosion lens system that works. You put it around a hollow supercritical mass, fire off the explosives, and you get a nuclear explosion. Well, maybe you get a full scale explosion and maybe the thing fizzles. Pop instead of BANG! The problem is that you are dependent upon having a spontaneous fission in the pit at the exact moment it is being compressed into a supercritical mass. Remember, the whole thing is in the process of exploding, and a few milliseconds after the pit reaches maximum compression it is going to bounce back and fly apart, and the fissile material will be scattered into thousands of tiny subcritical pieces without exploding. The critical mass of U235 is 52 Kg and that generates only about 117 spontaneous fissions per second, or one every 8,500 microseconds. The imploding pit remains at maximum compression less than 1 microsecond, so the chances are pretty good (8,500:1) that a spontaneous fission won't happen at the right time to start a chain reaction. You just can't count on fate kicking in a spare neutron at exactly the right moment.
To improve the probability that a chain reaction will start at the right time a variety of techniques have been used to generate a flood of neutrons in the pit just as it reaches maximum compression. In early bombs small neutron generators called urchins were placed at the center of the hollow pit. These contained two elements that released neutrons when they came together. The collapsing pit squashed these devices and caused the neutron generating elements to mix, releasing a burst of neutrons. The urchins were extremely radioactive, making them hazardous to handle and difficult to construct.
Later designs, starting with the Talos W-30 warhead, use neutron generators called zippers. These are small vacuum tube devices containing an ion source and electrodes for accelerating the ions. The ions are accelerated toward a target inside the tube. When the ions strike the target they cause a reaction that releases neutrons. These devices have the great advantage that they can produce a neutron flux on demand.
Neutron generators are placed outside the explosive lens and pit assembly, far enough away that the shock wave from the detonating high explosives will not reach them until after they have released their bursts of neutrons. This doesn't have to be very far away, because the shock wave travels through air at the speed of sound and over 25 times the speed of sound through the explosives. The implosion of the pit takes about 20-40 microseconds, and the shock wave from the explosives travels less than half an inch (1 centimeter) through air in this time.
The triggering mechanism for the warhead schedules firing of the neutron generators to produce a high neutron flux just as the pit is undergoing maximum compression by the implosion. The flood of neutrons initiates multiple chain reactions, ensuring that fissioning starts at the correct instant, and significantly improving the yield over what would result from a single spontaneous fission in the pit.
Tamper
A tamper is a container for the nuclear explosion. It is a shell of material that surrounds the pit, adding mass to the pit. At first the idea of the tamper sounds absurd - nothing can contain a nuclear explosion! However, the tamper doesn't have to contain the explosion very long. In a nuclear explosion all chain reaction fission events occur within a period of about half a microsecond (0.0000005 seconds)!
The fission chain reaction starts relatively slowly but increases exponentially - the faster it goes, the faster it goes. Each individual fission of a nucleus releases a small amount of energy, but in a short amount of time a huge number of nuclei fission and the total amount of energy released is enormous. The released energy heats up the pit and very quickly the temperature rises to about 10,000,000° Celsius (18,000,000° Fahrenheit). Consequently the pressure inside the pit increases drastically to millions of atmospheres, and starts to push back against the force of the implosion. It doesn't take long for the force of the nuclear explosion to exceed the force of the far weaker chemical implosion, and the pit starts to expand. When it expands the density of the fissile material drops rapidly and the nuclear chain reaction stops.
Although the pressures inside the pit are enormous, all of the imploding mass has inertia that must be overcome to start the whole thing exploding outward. Some time is required to stop the implosion and release the nuclear explosion. To get full yield something like 50-55 successive fission generations are required, but 99.9% of the energy is released in the last 7 generations - over a time period of about 0.07 microseconds (70 nanoseconds or billionths of a second). Even a very short delay in the expansion of the pit will allow another generation or two of the chain reaction, significantly increasing the amount of pit material that undergoes fission. Since each generation sees about a doubling in neutron release and capture, even an extremely short delay is all that is necessary to double or quadruple the yield of the explosion. The more mass that has to be accelerated the longer it takes to accelerate it outward, even if the accelerating force is enormous.
The tamper also serves a second function. Even with the best design and manufacturing the implosion wave may not be perfectly spherical. The shock wave from the high explosive lens encounters a sharp transition as it passes from the explosives to the tamper. If a lower density material surrounds the pit the shock wave will pass through the tamper relatively slowly. As the wave proceeds inward it is focused into an ever decreasing diameter volume. This tends to reduce any asymmetry in the implosion wave. As the radius of the imploding shock wave decreases the energy is concentrated into smaller amounts of tamper material, effectively amplifying the force of the shock (the total force does not increase, but the energy per unit volume increases). When this wave reaches the high density fissile material in the pit it delivers an extremely sharp high pressure blow to the surface. This acts like a hammer to crush the hollow pit and collapse it into a small high density supercritical ball.
A variety of materials have been used as tampers, including U238. The tamper in the Talos W-30 warhead was a fairly thick beryllium shell. Because beryllium is a relatively low density metal (light weight) it made an effective tamper that didn't increase warhead weight as much as heavier materials. But beryllium also has another important property.
Reflector
A relatively high neutron flux inside the pit is essential for ensuring that the nuclear chain reaction progresses. Many of the neutrons produced in the fissioning material escape the pit and contribute nothing to the reaction. One way to enhance neutron density within the pit is to surround it with a neutron reflector and send some of the escaping neutrons back into the pit. Some materials reflect neutrons like a mirror, and surrounding the pit with a reflector improves the efficiency of the nuclear reaction. However, not all escaping neutrons are reflected back, and the time required for a neutron to get back into the core is relatively long, so reflected neutrons do not contribute much to the fission chain reaction.
Beryllium metal is a very good neutron reflector. In the W-30 warhead the beryllium tamper also serves as the reflector. Warheads that use other materials for the tamper, such as U238, usually have a beryllium shell inside the tamper to serve as a neutron reflector.
Boosting
Another trick to increase weapons yield is called boosting. A mixture of deuterium (H2) and tritium (H3) gasses is injected into the center of the hollow pit just prior to the firing of the explosives to start the implosion. This gas is compressed at the center of the imploding pit. When the nuclear explosion occurs the temperatures suddenly rise high enough (about 10,000,000° Celsius) to cause the compressed deuterium and tritium to undergo fusion, where two nuclei of tritium and deuterium combine to produce a single helium nucleus. One or two free neutrons are generated by each fusion reaction. These free neutrons, produced at the center of the fissile material, cause additional fission reactions in the uranium or plutonium. These additional fissions significantly increase (boost) the amount of energy released before the pit is blown apart by the forces of the explosion.
Fusion of hydrogen to produce helium is the basis for thermonuclear weapons, which release hundreds or thousands of times as much energy as fission based nuclear weapons. However, in the boosting process a relatively small amount of energy is released by the fusion reaction, only a few percent of the total energy. But the free neutrons that are generated can double the number of uranium or plutonium atoms that fission (up to 40% of the fissile material), doubling the yield of the explosion.
One drawback to boosting is that tritium is very expensive to produce. However, boosting allows much less fissile material to be used, reducing the size and weight of the warhead, and fissile material is also very expensive. Tritium has a half life of 12.3 years so it decays 5.5% per year. This means the tritium reservoir in boosted weapons has to be replaced periodically to ensure the desired yield will be produced.
Physics Package
The collection of fissile material, reflector, tamper and explosive lens is sometimes referred to as the physics package. After the fissionable material is machined to the correct shape it is filled with an inert gas (typically nitrogen), surrounded by a shell of beryllium, and sealed to keep out moisture and oxygen. Then this pit is surrounded by a spherical shell of explosive lens elements. Early implosion designs used a collection of 32 elements arranged in the same pattern as the facets of a soccer ball (12 with 5 sides and 20 with 6 sides). Other designs were used with many more lens elements to get a more uniform spherical implosion. However, greater numbers of elements complicate the triggering process. All of the detonators must go off within an extremely short period of time, making the production, assembly, wiring and timing of the firing circuit more complex. Modern designs use far fewer lens elements and detonators.
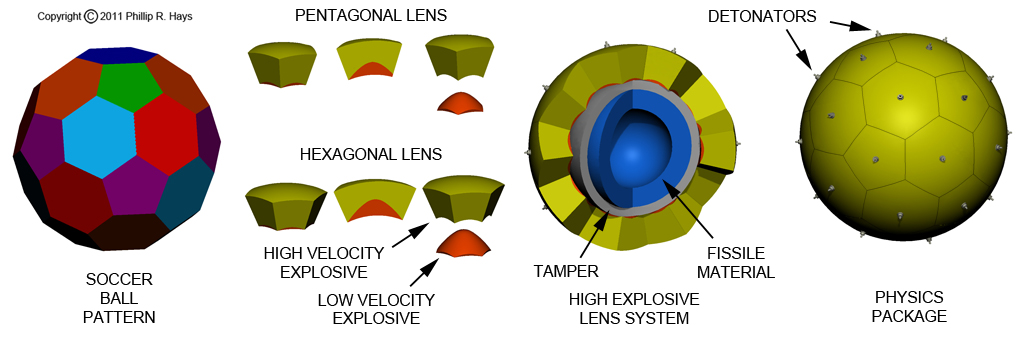
The Talos W-30 warhead was a 32 element assembly similar to what is shown in the picture above. It was the smallest nuclear warhead design at the time, just 22 inches diameter. The fissile material was Oralloy, a highly enriched uranium alloy (94% U235, 5% U238 and 1% U234). It was one of the first boosted production warheads, and it was the first to use external "zipper" neutron generators. The design contained many features to reduce size and weight, and to minimize the amount of very expensive fissile material. Key design elements were the neutron reflector, tamper, boosting, plastic explosive lens and external neutron generators. It represented the state of the art of nuclear weapons design in the mid 1950s.
Firing Process
When a nuclear weapon is detonated a carefully timed sequence of events has to occur. These may include encoded messages from the launch control to verify that weapon activation is authorized. First the power source must be turned on. Most weapons use a thermal battery that is inert during storage. Prior to arming the warhead a flammable "candle" is ignited to melt the electrolyte and charge the battery. Then the arming circuits are activated. The arming sequence may also include a collection of safety switches and environmental sensing to ensure that the warhead has been properly launched, dropped, etc. A short time prior to detonation the firing set is energized to build up the proper voltage for firing the detonators. At this time the deuterium and tritium booster gasses are transferred from the gas reservoir to the interior of the pit.
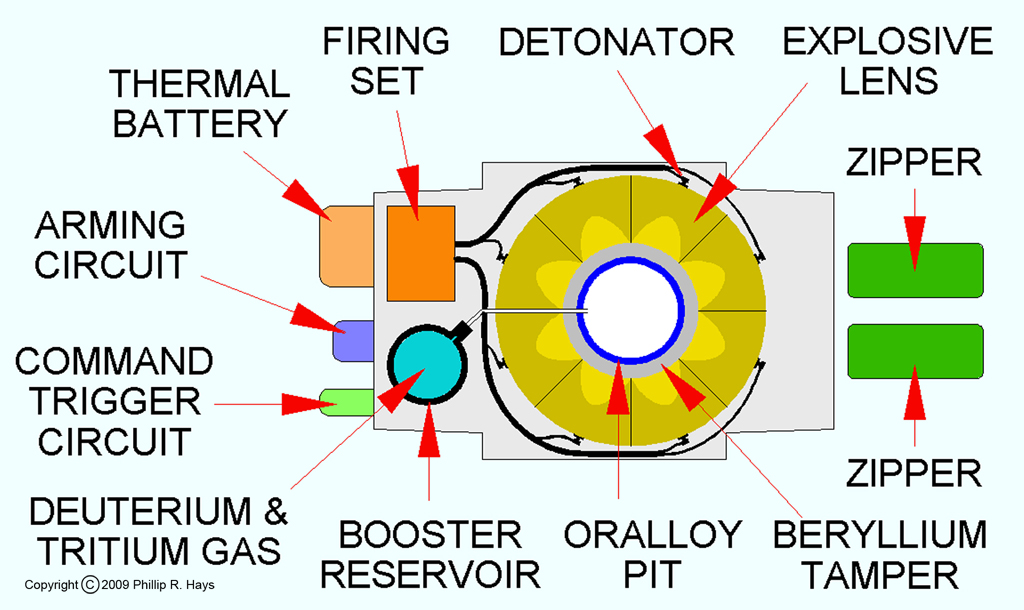
When the warhead receives the firing command a signal triggers the switch that transfers the electrical charge from the firing set to the detonators. This switch is designed to transfer the charge from the firing set to each of the detonator circuits at the same instant. The functioning of this switch is a critical part of nuclear weapons design. The cables from the switch to the detonators are cut to precise length so the charge will arrive at all of the detonators at exactly the same time, causing them to fire simultaneously. This is a critical part of the firing sequence. If any of the detonators fail, or fire too early or late, the explosion will be a dud, producing little or no nuclear yield. This is also a safety point for implosion warhead designs - they will not produce a significant nuclear explosion if a single detonator fires because of an accident.
When the detonators fire the explosive lens system produces a spherical implosion shock front that compresses the pit, causing it to become smaller and higher density. Implosion takes only 20 to 50 microseconds to achieve maximum compression, and this state lasts less than a microsecond. A timing circuit triggers the external neutron generator devices (zippers) just as the pit reaches maximum supercritical density. The flood of neutrons initiates many fission reactions in the fissile material, starting the nuclear chain reaction. Within half a millionth of a second a huge amount of energy is released, raising the temperature in the pit to about 10,000,000° Celsius. The extreme temperature causes the deuterium and tritium compressed at the center of the pit to undergo fusion, releasing a second flood of neutrons. These induce more fission reactions in the remaining fissile material, causing still more chain reactions. The reflector material sends many escaping neutrons back into the pit, further increasing the amount of fission.
Pressure inside the imploding pit suddenly increases drastically, overcoming the inertia of the imploding mass and forcing the warhead materials to start expanding. The mass of the tamper delays expansion briefly, allowing the nuclear chain reactions to proceed a bit longer, further increasing the yield of the explosion. But within a few millionths of a second after the initial nuclear fissions the warhead begins expanding rapidly. Fission products and the extreme temperatures quench the chain reaction and the extreme radiation causes secondary nuclear reactions in the remaining fissile material. The pit material density decreases rapidly and the remaining unspent fissile material becomes subcritical and fissioning stops. The sequence of events of the nuclear explosion is over in less than a millionth of a second.
The first external sign of the nuclear explosion is an intense burst of X-rays and gamma radiation, followed by intense neutron radiation. This radiation ionizes the atmosphere around the warhead and generates an initial brief flash of light from the explosion. Heat from this intense radiation vaporizes the entire warhead assembly and anything nearby. The energy of the nuclear reaction then blasts out and expands into an intensely bright nuclear fireball. The initial brief flash from the ionizing radiation followed by the longer flash of the fireball gives nuclear explosions a unique characteristic that allows them to be distinguished from other types of explosions. This feature is used by observation satellites to detect clandestine atmospheric nuclear explosions.
The fireball expands rapidly for about a second to a diameter of about two thousand feet for a small nuclear explosion. The heat from the flash is intense enough to ignite fires for a considerable distance. The blast wave from the explosion races outward from the fireball. The heat and blast cause most of the damage from the explosion. The initial radiation may cause some damage but exposure drops off rapidly with distance from the explosion. Anything that might suffer from the initial radiation burst will be destroyed by the heat and blast effects. The nuclear explosion also causes a sudden intense electromagnetic pulse that radiates outward for a considerable distance. This pulse can damage or destroy electronic equipment at a range of many miles.
Most of the damage from a small nuclear blast (20 kilotons or less) is over in a few seconds. The fireball cools sufficiently in three seconds that thermal radiation (heat) is no longer dangerous. Ten seconds after the blast the shock wave is about 2 1/2 miles from the point of explosion and wind velocity is about 40 miles per hour with an overpressure of about one pound per square inch, enough to cause minor damage to structures, ships or aircraft. In less than ten minutes the radioactive cloud rises to greater than 70,000 feet into the atmosphere.
Fallout
The fireball from a nuclear explosion contains all of the unexploded nuclear material from the pit, plus the initial fission products and structural materials in the warhead. All of these materials, plus nearby gasses in the atmosphere, are exposed to an intense flux of neutrons from the explosion. Just as uranium and plutonium nuclei can capture neutrons, the nuclei of all these other materials also capture neutrons and become new isotopes. Most of these isotopes are extremely radioactive, with half lives much shorter than a second. However some of the isotopes have half lives from minutes to years. All of these highly radioactive elements are carried in the fireball.
As hot materials in the cloud cool and condense the radioisotopes begin to drop out as rain or dust, creating radioactive "fallout." Within twelve hours after the explosion about 95% of the fission products have decayed to non-radioactive isotopes, but the remaining 5% can be dangerously radioactive, some of it for decades. This fallout leaves a radioactive trail downwind from the explosion. Atmospheric conditions determine where the fallout intensity will be greatest. Rain showers can wash much of the radioactive materials out of the cloud and deposit them in a relatively small area, resulting in a highly concentrated radioactive fallout.
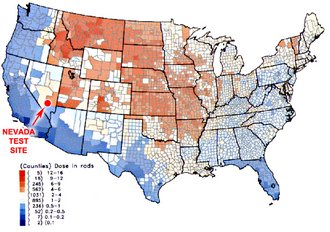
Fallout from the airburst tests at the Nevada test range typically blew east and northeast into the midwestern United States and southern Canada. Fallout intensity was not evenly distributed along the paths, but tended to be localized in areas where it rained. Significant amounts of radiation fell in many states, including places as far away from Nevada as Maine and North Carolina. The highest levels of fallout radiation exposure were equivalent to about 32 dental XRAYs. Even though this is not a high dose it is much greater than normal background radiation, and long term exposure poses health risks. Estimates of long term fatalities from radiation in this fallout range up to 80,000 people in the United States. That is about equal to American military combat fatalities in Korea and Vietnam. We killed as many of our own people as did our enemies.
In the event of a nuclear war the effects of fallout would be far more severe. Counterforce attacks would use ground bursts on underground targets like missile silos. Ground bursts expose far more material to intense neutron radiation, and the resulting fireball and cloud would carry much more material into the atmosphere and produce huge amounts of highly radioactive fallout. A study1 of the effects of an attack on a missile complex near Kansas City, Missouri, concluded that 150 one megaton surface bursts aimed at underground missile silos would destroy every structure that was not reinforced to withstand a nuclear blast in six counties, cause severe damage over another five counties, and produce a fallout cloud that would cover all of the State of Missouri. Within the blast areas fallout would be as high as 100,000 Roentgens. Directly downwind for 100 miles fallout intensity would be 25,000 Roentgens or greater, and 200 miles all the way across the State in Saint Louis fallout would be 10,000 Roentgens. Almost all of the State would receive a dose of 500 Roentgens or more for a period of one month.
Because different individuals respond to radiation differently, radiation dosages are stated in terms of the effect on populations. Scientists measure dosage of radiation (poisons, antibiotics, etc.) in terms of the dosage necessary to kill half of a population. This is called the "Lethal Dose 50%" or LD50. The LD50 for human populations is about 450 Roentgens. In other words, almost all of Missouri would be covered with fallout radioactive enough to kill more than half the population, and downwind areas would receive 20 to 50 times the LD50. In about two thirds of the State (including all large population areas) people would be incapacitated almost immediately after exposure and all would die in less than a day. In the rest of the State less than half the population would survive, and survivors would be convalescent for up to six months.
This study considered only one target, and considered the effects in only one State. Every major city and military base would be targets in a nuclear war, and the resulting fallout would cover the nation (and eventually winds would spread it over the entire planet). Clearly, the fallout from nuclear explosions is far more dangerous than the immediate damage from the blasts.
References
1. The Local Environment Resulting From A Massive Nuclear Attack On Whiteman Air Force Base, Final Technical Report SPC 537, Hurlburt, C. W., Yutko, C. A. and Sullivan, R. J., Federal Emergency Management Agency, Washington, D. C., April 1980.
